This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
Non-Medullary Thyroid Cancer & FOXE1 Gene
Non-Medullary Thyroid Cancer (NMTC) is a form of thyroid cancer that originates from the follicular cells of the thyroid gland. NMTC tends to be more prevalent in females than males and those with a family history of thyroid disease. While common symptoms of NMTC include persistent hoarseness or voice change, constant cough not due to cold, trouble swallowing or breathing, pain or vague discomfort in front of the neck, and a swelling or lump in the neck, most thyroid cancers typically do not present noticeable symptoms.
While the specific causes of NMTC are unknown, FOXE1 mutations have been associated with NMTC. FOXE1 is a thyroid transcription factor that regulates the transcription of DNA into RNA and belongs to a larger class of transcription factors known as the forkhead box protein family. Proteins within this family are characterized as being pioneer factors: transcription factors that can bind directly to condensed chromatin and can positively/negatively affect transcription. FOXE1 in particular is localized within the nucleus of the cell and regulates cell migration and cell differentiation through transcriptional regulation.
FOXE1 turns out to be highly conserved among different species. Not only that, the length of the protein as well as the position of their characteristic forkhead box (FH) protein domain is well conserved. While this FH protein domain plays an integral role sequence specific DNA binding, studies so far have only been able to show that mutations in this FH protein domain are associated with congenital hypothyrodism and thyroid dysgenesis (which is opposite of the characteristics of cancer). On the other hand, mutations in the low complexity region (LCR) of FOXE1 has shown association to an increased likelihood of NMTC and an upregulation of the WNT5A gene: a gene whose protein products have been associated with embryonic development and oncogenesis. This suggests that while the FH protein domain is essential for the function of FOXE1, the LCRs of FOXE1 also play an important role in the activity and mechanism of FOXE1.
Though FOXE1 is highly conserved among different species, Mus musculus was identified as an ideal model organism for their similarity to humans in anatomy, physiology, genomics, and diseased state phenotype. As NMTC subtypes are classified according to their histopathological characteristics, preexisting literature demonstrating the phenotypic similarities between mice and human NMTC subtypes helped make this decision. Figures [1]- [6].
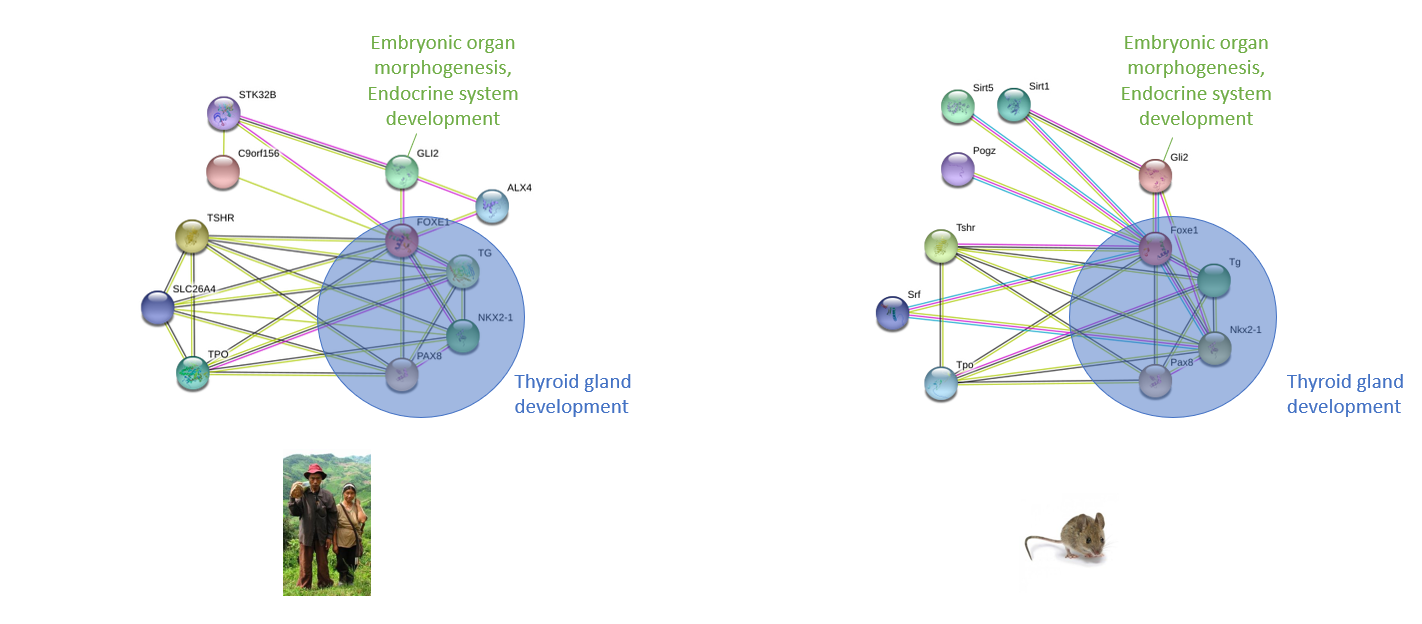
In addition to their phenotypic similarities, the similarity of the protein-protein interactions of FOXE1 in mice and humans helped decide mice as an ideal model organism. Figure [7]
While the specific causes of NMTC are unknown, FOXE1 mutations have been associated with NMTC. FOXE1 is a thyroid transcription factor that regulates the transcription of DNA into RNA and belongs to a larger class of transcription factors known as the forkhead box protein family. Proteins within this family are characterized as being pioneer factors: transcription factors that can bind directly to condensed chromatin and can positively/negatively affect transcription. FOXE1 in particular is localized within the nucleus of the cell and regulates cell migration and cell differentiation through transcriptional regulation.
FOXE1 turns out to be highly conserved among different species. Not only that, the length of the protein as well as the position of their characteristic forkhead box (FH) protein domain is well conserved. While this FH protein domain plays an integral role sequence specific DNA binding, studies so far have only been able to show that mutations in this FH protein domain are associated with congenital hypothyrodism and thyroid dysgenesis (which is opposite of the characteristics of cancer). On the other hand, mutations in the low complexity region (LCR) of FOXE1 has shown association to an increased likelihood of NMTC and an upregulation of the WNT5A gene: a gene whose protein products have been associated with embryonic development and oncogenesis. This suggests that while the FH protein domain is essential for the function of FOXE1, the LCRs of FOXE1 also play an important role in the activity and mechanism of FOXE1.
Though FOXE1 is highly conserved among different species, Mus musculus was identified as an ideal model organism for their similarity to humans in anatomy, physiology, genomics, and diseased state phenotype. As NMTC subtypes are classified according to their histopathological characteristics, preexisting literature demonstrating the phenotypic similarities between mice and human NMTC subtypes helped make this decision. Figures [1]- [6].
In addition to their phenotypic similarities, the similarity of the protein-protein interactions of FOXE1 in mice and humans helped decide mice as an ideal model organism. Figure [7]
|
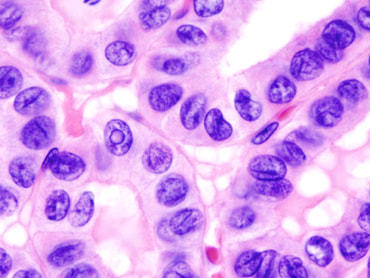
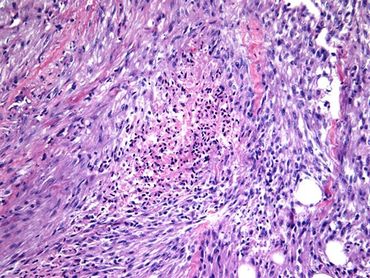
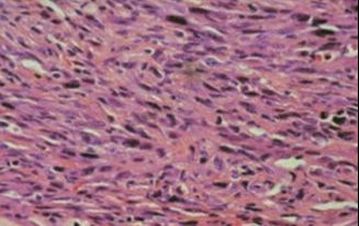
Figure [1]. Human PTC exhibiting ground-glass nuclei structure.
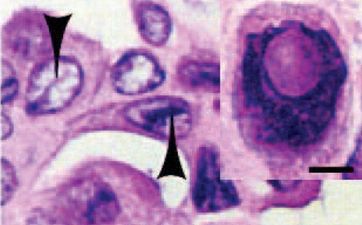
Figure [4]. Ret/PTC1 transgenic mouse PTC exhibiting ground-glass nuclei structure. [1]
|
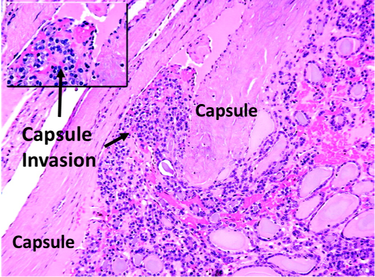
Figure [2]. Human FTC exhibiting capsular invasion.
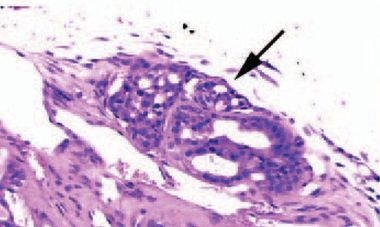
Figure [5]. TRβPV/- mouse FTC exhibiting capsular invasion. [1]
|
Figure [3]. Human ATC exhibiting poor cell differentiation.
Figure [6]. Mouse ATC exhibiting poor cell differentiation. [2]
|
Figure [7]. Protein-protein Interaction comparison of FOXE1 in humans and mice.
Aim 1: Identify conserved amino acids sequences that regulate cell growth/ proliferation
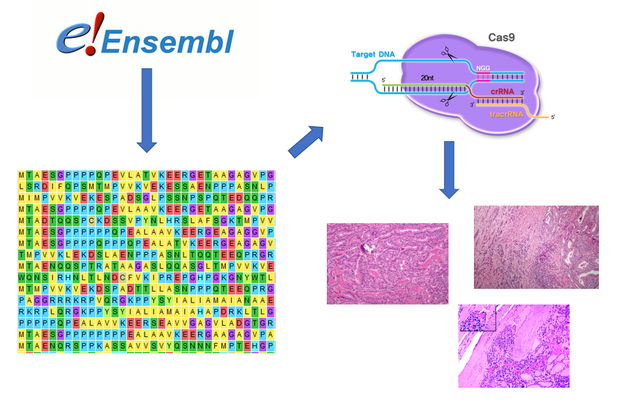
To identify conserved amino acid sequences in FOXE1 that are responsible for cell growth/ proliferation regulation, I first used Ensembl to retrieve the FASTA amino acid sequence files. After, the sequences were aligned using MEGA to identify conserved sequences. CRISPR- Cas 9 will then be utilized to make amino acid substitutions that are specific to species with thyroids. The model organisms (mice) will then be grown until maturity and screened for thyroid abnormalities.
The rationale behind this approach is that since different SNPs within the FOXE1 gene have been associated with different subtypes of NMTC, introducing subtype specific human -NMTC associated SNPs to homologous sequences within model organisms will allow observation of the acute effects of FOXE1 mutations. My hypothesis is the mice with mutated conserved FOXE1 amino acids specific to species with thyroid glands will lead to thyroid abnormalities.
The rationale behind this approach is that since different SNPs within the FOXE1 gene have been associated with different subtypes of NMTC, introducing subtype specific human -NMTC associated SNPs to homologous sequences within model organisms will allow observation of the acute effects of FOXE1 mutations. My hypothesis is the mice with mutated conserved FOXE1 amino acids specific to species with thyroid glands will lead to thyroid abnormalities.
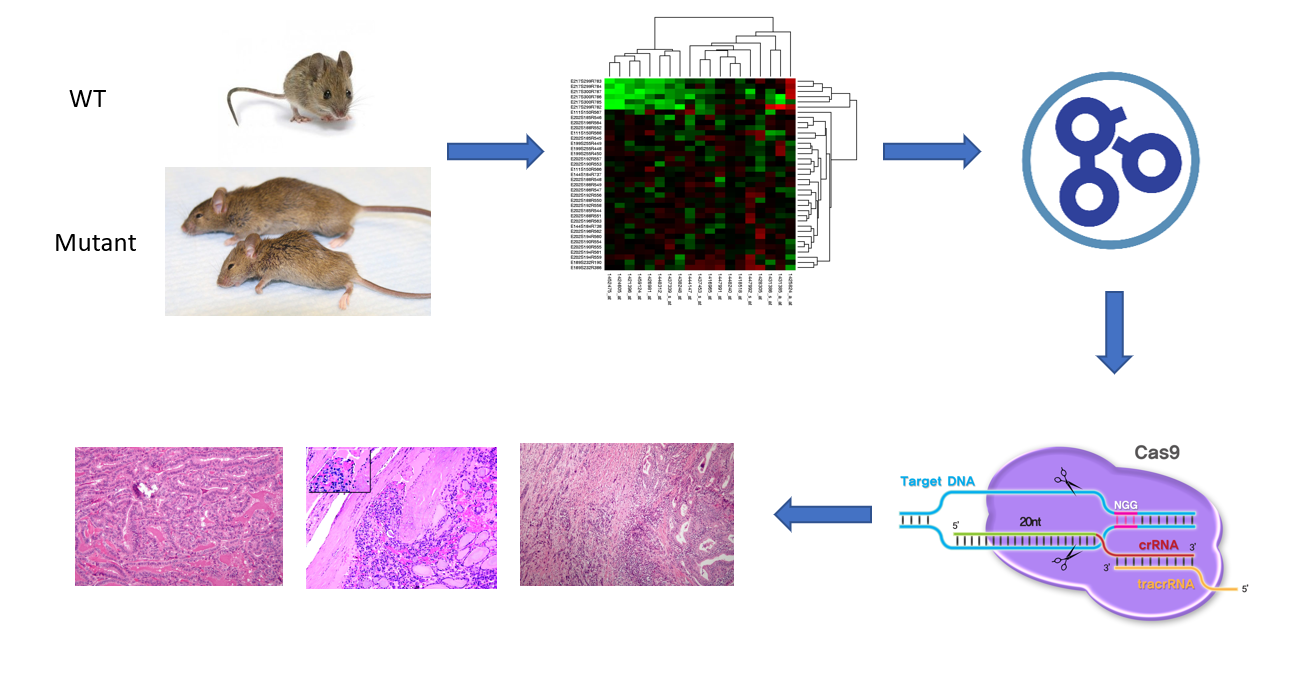
Figure [8]. Outline of Aim #1 workflow
Aim 2: Identify genes involved in thyroid morphogenesis
To identify genes involved in thyroid morphogenesis, follicular cells will be isolated from wild type (WT) and mutant (MT) FOXE1 mice. These isolated cells will then be put through single-cell RNA-seq and the data will be sorted using Gene Ontology (GO) terminology and compared between the WT and MT mice. Changes in molecular functions will be searched for. Differentially expressed genes will then either be knocked out or rendered inactive in mice using CRISPR- Cas 9. The mice will then be screened to confirm the involvement of differentially expressed genes in thyroid morphogenesis.
The rationale for this approach is that by analyzing the RNA-seq data and GO analysis between WT and MT mice, we can determine which mutations are associated with changes in expression levels of genes associated with thyroid morphogenesis. My hypothesis is that genes responsible for molecular functions such as growth factor binding will be over or under expressed in FOXE1 mutant mice.
The rationale for this approach is that by analyzing the RNA-seq data and GO analysis between WT and MT mice, we can determine which mutations are associated with changes in expression levels of genes associated with thyroid morphogenesis. My hypothesis is that genes responsible for molecular functions such as growth factor binding will be over or under expressed in FOXE1 mutant mice.
Figure [9]. Outline of Aim #2 workflow
Aim 3: Characterize phosphorylation patterns of NMTC
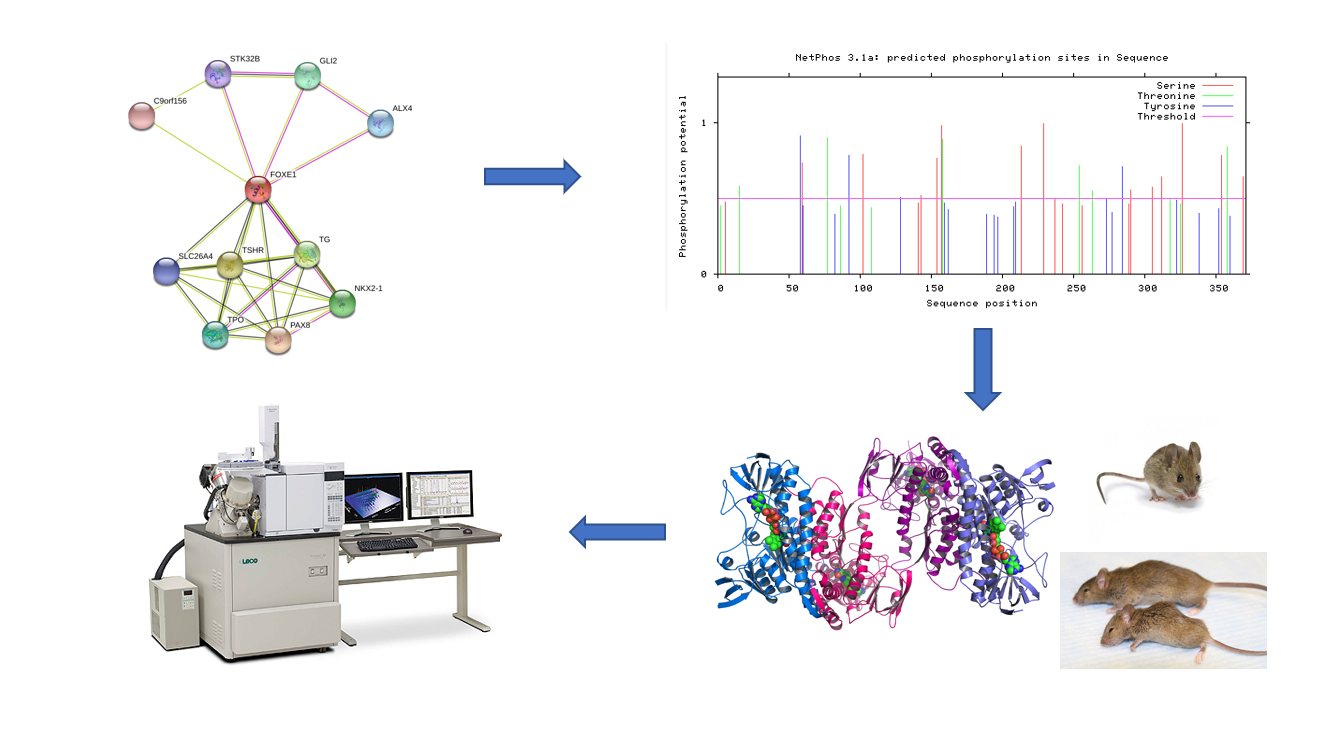
To characterize the phosphorylation patterns of NMTC, I will first identify proteins that interact with FOXE1 using STRING. After the FOXE1 interacting proteins are identified, NETPHOS will be used to identify putative phosphorylation sites for FOXE1 and FOXE1 interacting proteins. Then, a phosphoproteomic study will be performed on the thyroid follicular cells of WT and MT FOXE1 mice using mass spectrometry. The phosphorylation patterns will then be compared and differences between the WT and MT will be searched for.
The rationale for this approach is that since cell growth and proliferation are dependent on a variety of cell signalling pathways, FOXE1 may alter the normal cell signalling pathways by either activation or deactivation of certain signalling pathways via phosphorylation. My hypothesis is that proteins responsible for cell growth/ proliferation signallling pathways will be over- or under- phosphorylated.
The rationale for this approach is that since cell growth and proliferation are dependent on a variety of cell signalling pathways, FOXE1 may alter the normal cell signalling pathways by either activation or deactivation of certain signalling pathways via phosphorylation. My hypothesis is that proteins responsible for cell growth/ proliferation signallling pathways will be over- or under- phosphorylated.
Figure [10]. Outline of Aim #3 workflow
Future Directions
FOXE1 plays an important role in regulating thyroid cell growth and proliferation. A better understanding of FOXE1's regulatory roles and mechanisms will allow for a better understanding of NMTC as a whole and potentially help understand the regulatory mechanisms involved in other types of cancers. Similar studies regarding transcription factors of different cell types may ultimately help identify ways to stop the progression of NMTC as well as other types of cancer.
Figure [11]. General outline of cancer progression
|
Final Presentation
|
Final Presentation Draft
| ||||||||||||||||||
Figures
[1] K. A. B. Knostman, S. M. Jhiang, and C. C. Capen (2007). Genetic Alterations in Thyroid Cancer: The Role of Mouse Models. Veterinary Pathology, Vol 44, Issue 1, pp. 1 - 14. https://doi.org/10.1354/vp.44-1-1
[2] Champa, D., & Di Cristofano, A. (2015). Modeling anaplastic thyroid carcinoma in the mouse. Hormones & Cancer, 6(1), 37–44. http://doi.org/10.1007/s12672-014-0208-8
[3] https://thedotingskeptic.files.wordpress.com/2016/02/crispr3.jpg
[4] https://upload.wikimedia.org/wikipedia/commons/6/65/Papillary_thyroid_carcinoma_tall_cell_var_intermed_mag.jpg
[5] https://en.wikipedia.org/wiki/Anaplastic_thyroid_cancer#/media/File:Anaplastic_thyroid_carcinoma_low_mag.jpg
[6] http://jcp.bmj.com/content/jclinpath/60/3/244/F2.large.jpg
[7] https://www.sperietori.ro/images/produse/SoareceleZdeZcasaZMusZMusculus_b_1.jpg
[8] https://www.researchgate.net/profile/Ken_Yamashita3/publication/266625227/figure/fig1/AS:203126704480260@1425440588553/Upper-wild-type-mouse-lower-Klotho-mutant-mouse-The-Klotho-mouse-is-smaller-in-size.png
[9] https://s3.amazonaws.com/go-public/image/go-logo.large.png
[10] https://commons.wikimedia.org/wiki/File:Heatmap.png
[11] http://www.cbs.dtu.dk/services/NetPhos/
[12] http://www.tuscany-diet.net/wp-content/uploads/2015/04/protein-structure.jpg
[13] http://www.consulente-energia.com/iz113.jpg
[14] https://string-db.org/
[15] https://thumbs.dreamstime.com/z/process-cancer-cell-development-medical-illustration-woman-touching-her-head-has-headaches-fever-72531182.jpg
[1] K. A. B. Knostman, S. M. Jhiang, and C. C. Capen (2007). Genetic Alterations in Thyroid Cancer: The Role of Mouse Models. Veterinary Pathology, Vol 44, Issue 1, pp. 1 - 14. https://doi.org/10.1354/vp.44-1-1
[2] Champa, D., & Di Cristofano, A. (2015). Modeling anaplastic thyroid carcinoma in the mouse. Hormones & Cancer, 6(1), 37–44. http://doi.org/10.1007/s12672-014-0208-8
[3] https://thedotingskeptic.files.wordpress.com/2016/02/crispr3.jpg
[4] https://upload.wikimedia.org/wikipedia/commons/6/65/Papillary_thyroid_carcinoma_tall_cell_var_intermed_mag.jpg
[5] https://en.wikipedia.org/wiki/Anaplastic_thyroid_cancer#/media/File:Anaplastic_thyroid_carcinoma_low_mag.jpg
[6] http://jcp.bmj.com/content/jclinpath/60/3/244/F2.large.jpg
[7] https://www.sperietori.ro/images/produse/SoareceleZdeZcasaZMusZMusculus_b_1.jpg
[8] https://www.researchgate.net/profile/Ken_Yamashita3/publication/266625227/figure/fig1/AS:203126704480260@1425440588553/Upper-wild-type-mouse-lower-Klotho-mutant-mouse-The-Klotho-mouse-is-smaller-in-size.png
[9] https://s3.amazonaws.com/go-public/image/go-logo.large.png
[10] https://commons.wikimedia.org/wiki/File:Heatmap.png
[11] http://www.cbs.dtu.dk/services/NetPhos/
[12] http://www.tuscany-diet.net/wp-content/uploads/2015/04/protein-structure.jpg
[13] http://www.consulente-energia.com/iz113.jpg
[14] https://string-db.org/
[15] https://thumbs.dreamstime.com/z/process-cancer-cell-development-medical-illustration-woman-touching-her-head-has-headaches-fever-72531182.jpg
Image References
[1] http://goodthinkinc.com/wp-content/uploads/2014/06/brain.gears_.thinking.jpg
[1] http://goodthinkinc.com/wp-content/uploads/2014/06/brain.gears_.thinking.jpg